A) 90°
B) 109.5°
C) 120°
D) 107°
E) 105°
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization on the central atom in NO3- ?
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular geometry of N2O as predicted by the VSEPR model? 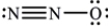
A) trigonal pyramidal
B) trigonal planar
C) tetrahedral
D) bent
E) linear
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is true concerning the dipole moment of a CO2 molecule?
A) The dipole moment is zero because the C-O bond is nonpolar.
B) The dipole moment is zero because the molecule is linear.
C) The dipole moment is zero because the molecule is bent.
D) The dipole moment is nonzero because the molecule is linear.
E) The dipole moment is nonzero because the molecule is bent.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following molecules in order of increasing dipole moment. 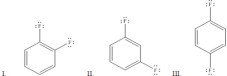
A) III < II < I
B) III < I < II
C) I < II < III
D) I < III < II
E) II < III < I
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular geometry of the thiocyanate anion,SCN-,as predicted by the VSEPR model? (Carbon is the central atom.)
A) linear
B) bent
C) tetrahedral
D) trigonal planar
E) trigonal pyramidal
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules is polar?
A) PBr5
B) CCl4
C) BrF5
D) XeF2
E) XeF4
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Valence bond theory predicts that sulfur will use _____ hybrid orbitals in sulfur dioxide,SO2 .
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to molecular orbital theory,what is the bond order in the O2- ion?
A) 5.5
B) 5
C) 4
D) 2.5
E) 1.5
G) C) and E)
Correct Answer

verified
E
Correct Answer
verified
Multiple Choice
Hyposulfite,SO22-,is a polyatomic ion that has not yet been definitively confirmed to exist.However,the VSEPR model may still be applied to yield a prediction on its molecular geometry.What is the most reasonable estimate for the O-S-O bond angle in hyposulfite?
A) 90°
B) 106°
C) 109.5°
D) 112°
E) 120°
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization of the As atom in the AsF5 molecule?
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the VSEPR model,the predicted molecular geometry of the SO3 molecule is
A) pyramidal.
B) tetrahedral.
C) trigonal planar.
D) seesaw.
E) square planar.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Water has ___________ (number)lone pair(s)of electrons on the central oxygen.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using the VSEPR model,predict the molecular geometry around the central atom in PO43-.
A) trigonal planar
B) trigonal pyramidal
C) tetrahedral
D) trigonal bipyramidal
E) octahedral
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the VSEPR model,a molecule with the general formula AB5 with one lone pair on the central atom will have a ______ molecular geometry.
A) tetrahedral
B) trigonalbipyramidal
C) square pyramidal
D) octahedral
E) seesaw
G) All of the above
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Allene,C3H4 (shown below) ,is one component of gas used for high-temperature welding. According to the valence bond model,which is a proper description of the bonding on the central carbon atom in allene? 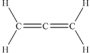
A) It shares four π bonds,two with each carbon atom on either side.
B) It shares four σ bonds,two with each carbon atom on either side.
C) It shares one σ bond and one π bond with each carbon atom on either side.
D) It shares two σ bonds with the carbon atom on the left,and two π bonds with the carbon atom on the right.
E) It shares two σ bonds with the carbon atom on the right,and two π bonds with the carbon atom on the left.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Thiocarbonyl disulfide (CSF2) ,based on the coordinate axes provided below,in which direction does the net molecular dipole moment point for the molecule? 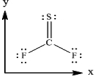
A) It points in the +x direction.
B) It points in the -x direction.
C) It points in the +y direction.
D) It points in the -y direction.
E) The molecule does not have a net molecular dipole moment.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular geometry of ClF2- as predicted by the VSEPR model?
A) linear
B) bent
C) seesaw
D) T-shaped
E) trigonal bipyramidal
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which formula is incorrectly matched with its VSEPR model representation? (Note: Lone pairs on the models,if any,are not shown.)
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) C) and D)
Correct Answer

verified
A
Correct Answer
verified
True/False
The BrF5 molecule has polar bonds and has a net dipole moment.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 137
Related Exams