A) paramagnetic,with 1 unpaired electron.
B) paramagnetic,with 3 unpaired electrons.
C) paramagnetic,with 4 unpaired electrons.
D) paramagnetic,with 5 unpaired electrons.
E) diamagnetic.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a coordination compound involving a complex ion of square planar geometry,which of the following types of isomerism is/are never possible?
A) Geometric
B) Optical
C) Linkage
D) Coordination
E) More than one of the choices is correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why are phosphates being eliminated from detergents?
A) Detergents with phosphates are less efficient cleansers.
B) By chelating Ca2+ ions,the phosphates allow more efficient cleansing.
C) Phosphates are important plant nutrients which increase algae growth and deplete oxygen.
D) None of the.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following octahedral complexes should have the largest crystal field splitting energy,Δ?
A) [Cr(H2O) 6]3+
B) [Cr(SCN) 6]3
C) [Cr(NH3) 6]3+
D) [Cr(CN) 6]3-
E) [Cr(en) 3]3+ (en = ethylenediamine)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the spectrochemical series,which one of the following ligands has the strongest field?
A) H2O
B) CN-
C) NH3
D) OH-
E) Cl-
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Bidentate and polydentate ligands are called ___________ __________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the complex ion [Fe(CN) 6]4-,what is the oxidation number of Fe?
A) +1
B) +2
C) +3
D) -4
E) +6
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
A complex ion that undergoes a very slow exchange reaction is called an inert complex.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many 3d electrons does a ground-state Sc3+ ion have?
A) 1
B) 3
C) 2
D) 10
E) 0
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the electron configuration of a ground-state Cr atom?
A) 1s22s22p63s23p64s23d4
B) 1s22s22p63s23p63d6
C) 1s22s22p63s23p64s14d5
D) 1s22s22p63s23p64s24d4
E) 1s22s22p63s23p64s13d5
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
What terms describe the geometrical isomers that are possible for the complex [Pt(NH3)2Cl4]?
Correct Answer

verified
cis and trans
Correct Answer
verified
Multiple Choice
Which of the following metals is capable of forming complexes spanning the smallest range of oxidation numbers?
A) Cu
B) Sc
C) Fe
D) Mn
E) Ni
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ligands is most likely to form a low-spin octahedral complex with iron(III) ?
A) Cl-
B) H2O
C) NH3
D) OH-
E) CO
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
In complexes of transition metals,the maximum coordination number of the metal is equal to its number of d electrons.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The net rotation of plane-polarized light by a racemic mixture is _______.
A) to the right
B) to the left
C) zero
E) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
What is used to measure the rotation of polarized light as it passes through an atom?
Correct Answer

verified
Correct Answer
verified
Short Answer
One of the uses of EDTA is in the treatment of ________ ___________.
Correct Answer

verified
metal poisoning
Correct Answer
verified
Short Answer
In a reaction between a metal ion and a group of anions or a polar molecule,the metal ion acts as a ______ ________.
Correct Answer

verified
Lewis acid
Correct Answer
verified
Multiple Choice
Which name could correspond to the following coordination compound,where M represents a transition metal cation and L represents a ligand? 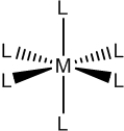
A) Hexachloroferrate(III)
B) Hexaammineiron(III)
C) Hexaaquochromium(II)
D) Hexacyanomanganate(II)
E) All of the are possible.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
What are the two types of stereoisomers that coordination compounds may exhibit?
Correct Answer

verified
geometrica...View Answer
Show Answer
Correct Answer
verified
View Answer
Showing 1 - 20 of 128
Related Exams