Correct Answer

verified
The outermost (ns2) e...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which of the following ions is most likely to form colored compounds?
A) Sc3+
B) Cu+
C) Zn2+
D) Cr3+
E) Ca2+
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the spectrochemical series, which one of the following ligands has the strongest field?
A) H2O
B) CN-
C) NH3
D) OH-
E) Cl-
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following will be diamagnetic?
A) Ni2+
B) Cr2+
C) Mn2+
D) Co3+
E) Ti4+
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
All atoms of the first transition series of elements have the ground state electronic configuration [Ar]4s23dx, where x is an integer from 1 to 10.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
All the actinide series of transition elements are radioactive.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to valence bond theory, what would be the set of hybrid orbitals used when a Period 4 transition metal forms a tetrahedral complex?
A) d2sp
B) dsp2
C) dsp3
D) sp3
E) d2p2
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the highest possible oxidation state for molybdenum, Mo?
A) +2
B) +4
C) +6
D) +8
E) none of the above
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many unpaired electrons are there in the Fe3+ ion?
A) 5
B) 4
C) 3
D) 2
E) 1
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The most common oxidation state for ions of the transition elements is
A) +2.
B) +3.
C) +4.
D) +5.
E) +6.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aluminum reacts with oxygen in the air to form a protective oxide coating. Silver also reacts with compounds in air to form a black coating. What substance is formed?
A) silver oxide
B) silver chloride
C) silver sulfide
D) silver carbonate
E) silver nitride
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following structures (1 and 2 are octahedral; 3 and 4 are square planar) . 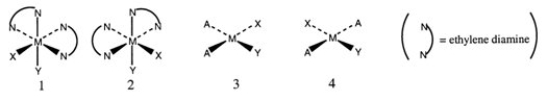 Which one of the following statements about the above structures is correct?
Which one of the following statements about the above structures is correct?
A) 1 and 2 are superimposable.
B) 1 and 2 are geometric isomers.
C) 3 and 4 are structural isomers.
D) 3 and 4 are optical isomers.
E) 3 and 4 are geometric isomers.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following transition elements can have an oxidation number of +7?
A) V
B) Cr
C) Mn
D) Fe
E) Co
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The compound K3[Fe(CN) 6] is used in calico printing and wool dyeing. Give its systematic name.
A) potassium iron(III) hexacyanate
B) tripotassium iron(III) hexacyanate
C) potassium hexacyanoferrate(III)
D) potassium hexacyanideferrate
E) none of the above
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If M represents a transition element, which of the following oxides should be the least basic?
A) MO
B) M2O
C) M2O3
D) MO2
E) MO3
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to valence bond theory, what would be the set of hybrid orbitals used when a Period 4 transition metal forms a square planar complex?
A) d2sp
B) d2p2
C) dsp3
D) sp3
E) dsp2
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ground state electronic configuration of Cr2+ is
A) [Ar]4s13d5.
B) [Ar]4s23d4.
C) [Ar]3d4.
D) [Ar]4s13d3.
E) [Ar]4s23d2.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to Valence Bond theory, in the square planar Ni(CN) 42- complex ion, the orbital hybridization pattern is
A) sp3.
B) dsp2.
C) d2sp.
D) d2sp3.
E) none of the above.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ions could exist in only the high-spin state in an octahedral complex?
A) Cr2+
B) Mn4+
C) Fe3+
D) Co3+
E) Ni2+
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Essay
The dxy and the dx2-y2 orbitals both lie in the xy plane, yet for a metal ion in an octahedral complex the energy of the dxy orbital is lower than that of the dx2-y2orbital. Explain this using the arguments of crystal field theory.
Correct Answer

verified
The dxy orbital lobes are directed between...View Answer
Show Answer
Correct Answer
verified
View Answer
Showing 41 - 60 of 92
Related Exams