A) The coordination number of the atoms in the lattice is 8.
B) The packing in this lattice is more efficient than for a body-centered cubic system.
C) If the atoms have radius r, then the length of the cube edge is 8 × r.
D) There are four atoms per unit cell in this type of packing.
E) The packing efficiency in this lattice and hexagonal close packing are the same.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In hydrogen iodide __________________ are the most important intermolecular forces.
A) dipole-dipole forces
B) London dispersion forces
C) hydrogen bonding
D) covalent bonds
E) polar covalent bonds
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances does not exist in the indicated solid type?
A) graphite - network
B) Na - metallic
C) SiO2 - molecular
D) NaCl - ionic
E) diamond - network
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Use molecular orbital band diagrams to explain why metals are good conductors but semiconductors are not.
Correct Answer

verified
 In metals, the conduction and valence b...
In metals, the conduction and valence b...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
When silicon is doped with an element from group 3A(13) , the device/material produced is a/an
A) intrinsic semiconductor.
B) p-type semiconductor.
C) n-type semiconductor.
D) p-n junction.
E) transistor.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the electron cloud of a molecule is easily distorted, the molecule has a high _____________.
A) polarity
B) polarizability
C) dipole moment
D) van der Waals radius
E) compressibility
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The strongest intermolecular interactions between hydrogen fluoride (HF) molecules arise from
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) ion-dipole interactions.
E) ionic bonds.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Essay
a. State the essential requirements for hydrogen bonding to be important in a compound. b. List four properties of water which are significantly influenced by the presence of hydrogen bonding.
Correct Answer

verified
a. The compound must contain hydrogen bo...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
A metal with a body-centered cubic lattice will have ______ atom(s) per unit cell.
A) 1
B) 2
C) 3
D) 4
E) 9
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Examine the following phase diagram and identify the feature represented by point A. 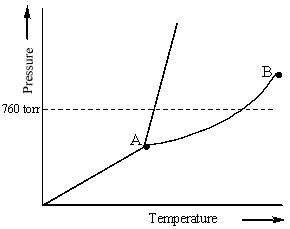
A) melting point
B) critical point
C) triple point
D) sublimation point
E) boiling point
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
The energy of a hydrogen bond is greater than that of a typical covalent bond.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is true about kinetic energy, Ek, and potential energy, Ep, when ethyl alcohol at 40°C is compared with ethyl alcohol at 20°C?
A) Ek(40°C) < Ek(20°C) ; Ep(40°C) Ep(20°C)
B) Ek(40°C) > Ek(20°C) ; Ep(40°C) Ep(20°C)
C) Ep(40°C) < Ep(20°C) ; Ek(40°C) Ek(20°C)
D) Ep(40°C) > Ep(20°C) ; Ek(40°C) Ek(20°C)
E) Ep(40°C) > Ep(20°C) ; Ek(40°C) > Ek(20°C)
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sample of octane in equilibrium with its vapor in a closed 1.0-L container has a vapor pressure of 50.0 torr at 45°C. The container's volume is increased to 2.0 L at constant temperature and the liquid/vapor equilibrium is reestablished. What is the vapor pressure?
A) > 50.0 torr
B) 50.0 torr
C) 25.0 torr
D) The mass of the octane vapor is needed to calculate the vapor pressure.
E) The external pressure is needed to calculate the vapor pressure.
G) C) and D)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Neon atoms are attracted to each other by
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) covalent bonding.
E) intramolecular forces.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Mercury melts at -39°C and boils at 357°C. Draw a diagram of the heating curve of mercury. Label all lines and axes, and clearly indicate the melting and boiling points on your diagram.
Correct Answer

verified
11ea8ef8_2b5e_fb68_ab0a_9bbc95341bc8_TB5833_00
Correct Answer
verified
True/False
A liquid may be made to boil at room temperature, simply by lowering the pressure.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
The vapor pressure of 1-butene is 1.268 atm at 273.15 K and its heat of vaporization is 22.9 kJ/mol. What is the normal boiling point of 1-butene?
Correct Answer

verified
266.9 K
Correct Answer
verified
Multiple Choice
Which of the following atoms should have the smallest polarizability?
A) Si
B) S
C) Te
D) Bi
E) Br
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Of the five major types of crystalline solid, which would you expect each of the following to form? (e.g., H2O: molecular) a. Sn b. Si c. KCl d. Xe e. F2
Correct Answer

verified
a. metallic
b. netwo...View Answer
Show Answer
Correct Answer
verified
b. netwo...
View Answer
Multiple Choice
Which of the following pairs is arranged with the particle of higher polarizability listed first?
A) Se2-, S2-
B) I, I-
C) Mg2+, Mg
D) Br, I
E) none of the above
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 111
Related Exams