A) n = 1
B) n = 2
C) n = 3
D) n = 4
E) n = 5
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A hydrogen atom is in a quantum state with principal quantum number n = 2.The atom absorbs a photon with a wavelength of 660 nm.Determine the maximum possible orbital angular momentum of the electron after absorption.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the longest possible wavelength that a hydrogen atom will emit?
A) 91 nm
B) 122 nm
C) 365 nm
D) 1459 nm
E) There is no limit.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What are the quantum numbers n and l for a hydrogen atom with E = - (13.6/16) eV and L = 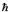 ?
?
A) n = 4,l = 2
B) n = 4,l = 6
C) n = 3,l = 3
D) n = 3,l = 6
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A hydrogen atom relaxes to a lower energy state by emitting a 2.55 eV photon.What is the lower energy state?
A) n = 1
B) n = 2
C) n = 3
D) n = 4
E) n = 5
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the wavelength of the photon emitted by a He+ when the electron jumps from the n = 7 to the n = 4 state?
A) 8.8 m
B) 3.4 m
C) 4.4 m
D) 896 nm
E) 542 nm
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the Bohr model of hydrogen,as the principle quantum number n increases,
A) the total energy increases and the kinetic energy decreases.
B) the potential energy increases and the speed decreases.
C) the orbital radius increases.
D) All are correct.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The energy contained in a length of a 3.00-mW laser beam in vacuum is 1.80 * 10-10 J.What is the length of the beam?
A) 2.0 m
B) 24 m
C) 15 m
D) 18 m
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electron in a singly ionized helium atom,which consists of two protons,two neutrons and only one electron,makes a transition from the n = 2 state to the n = 1 state,emitting a single photon in the process.What is the energy of this photon?
A) 3.40 eV
B) 10.2 eV
C) 13.6 eV
D) None are correct.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If the period of rotation of electron in the Bohr hydrogen atom is 4.10 fs,which Bohr orbit does the electron occupy?
A) first
B) second
C) third
D) fourth
E) fifth
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the magnitude (in N) of the electric force between a proton and a (lowest energy) n = 1 electron in a hydrogen atom?
A) 8.2 * 10-8
B) 8.2 * 108
C) 8.2 * 10-12
D) 8.2 * 10-4
E) 8.2 * 104
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The energy contained in laser beam of length 2.00 m that moves through vacuum is 1.80 * 10-10 J.What is the power of the laser?
A) 3.0 mW
B) 12 mW
C) 23 mW
D) 27 mW
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The frequency of the  line of the Lyman group for the hydrogen spectrum is
line of the Lyman group for the hydrogen spectrum is
A) 9.88 * 1014 Hz
B) 3.22 * 1015 Hz
C) 1.21 * 1015 Hz
D) 2.25 * 1015 Hz
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Pfund series results from emission/absorption of photons due to transitions of electrons in a hydrogen to/from the n=5 energy level from/to higher energy levels.What is the longest wavelength photon emitted in the Pfund series?
A) 658 nm
B) 2.29 µm
C) 2.74 µm
D) 7.47 µm
E) 12.4 µm
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Positronium consists of a bound state of an electron and a positron.A positron is exactly the same as an electron,except it has a charge of +e instead of -e.In the classical model,both particles orbit a point midway between them.The reduced mass of positronium is equal to
A) the mass of an electron.
B) the mass of an electron squared.
C) the square root of the mass of an electron.
D) None are correct.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 5.0 UV laser emits light that has a wavelength 350 nm.If 1.23 * 1018 photons are emitted in a pulse,what is the duration of the pulse?
A) 140 ns
B) 89 ns
C) 54 ns
D) 110 ns
E) 10 ns
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A collection of hydrogen atoms has been placed into the n = 4 excited state.Which of the following wavelengths of photons will NOT be emitted by the hydrogen atoms as they decay back to the ground state?
A) 97 nm
B) 490 nm
C) 970 nm
D) 1900 nm
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electrons are allowed to inhabit the M-shell of an atom?
A) 18
B) 10
C) 8
D) 32
E) 14
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the angular momentum of the orbiting electron in a hydrogen atom with quantum number of 45?
A) 0.4 * 10-31 kg m2/s
B) 1.6 * 10-31 kg m2/s
C) 2.3 * 10-33 kg m2/s
D) 4.7 * 10-33 kg m2/s
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The frequency of the 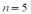 line of the Balmer group for the hydrogen spectrum is
line of the Balmer group for the hydrogen spectrum is
A) 6.91 * 1014 Hz
B) 7.33 * 1014 Hz
C) 9.05 * 1014 Hz
D) 8.54 * 1014 Hz
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 72
Related Exams