A) Ga
B) Zn
C) Cd
D) Hg
E) Pb
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Find the emf of the cell described by the cell diagram Fe | Fe2+ (1.500M) || Au3+ (0.00400M) | Au
A) 1.99 V
B) 1.89 V
C) 1.94 V
D) 1.66 V
E) 1.91 V
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the following half equation, which is the oxidizing agent? NO3-(aq) + 4H+(aq) + 3e- NO(g) + 2H2O
A) NO3-
B) H+
C) e-
D) NO
E) H2O
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Consider the reaction Fe + Sn2+(1 10-3 M) Fe2+(1.0 M)+ Sn. Sketch a diagram of such a cell. Be sure to label the anode and cathode, and show the direction of electron flow in the external circuit. Write the half-reactions occurring at the electrodes.
Correct Answer

verified
Fe | Fe2+(1.0 M)|| ...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which statement is true for a spontaneous redox reaction carried out at standard-state conditions?
A) E°red is always negative.
B) E°cell is always positive.
C) E°ox is always positive.
D) E°red is always positive.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Iron objects such as storage tanks and underground pipelines can be protected from corrosion by connecting them through a wire to a piece of
A) Pb
B) Ag
C) Sn
D) Mg
E) Cu
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these metals will be oxidized in hydrochloric acid under standard conditions at 25°C?
A) Ag
B) Au
C) Hg
D) Cu
E) Zn
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete and balance the following redox equation using the set of smallest whole-numbers coefficients. What is the sum of the coefficients? HI + HNO3 I2 + NO (acidic solution)
A) 5
B) 7
C) 14
D) 17
E) None of these.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Aluminum metal is formed by the electrolysis of Al2O3 in molten cryolite. How many minutes are required to form 10.0 g of Al using a current of 30 A?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reaction 2Fe3+(aq) + Fe(s) 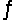 3Fe2+(aq) . Find the equilibrium constant for this reaction at 25°C.
3Fe2+(aq) . Find the equilibrium constant for this reaction at 25°C.
A) 1 1011
B) 8 1040
C) 3 1020
D) 7 10-12
E) 1 10-41
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Will H2(g)form when Ag is placed in 1.0 M HCl?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these metals will not reduce water to hydrogen in basic solution under standard conditions?
A) Cd
B) Sr
C) Mg
D) Ba
E) K
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Aluminum metal is formed by the electrolysis of Al2O3 in molten cryolite. How many grams of Al are produced when 6.50 103 C pass through the cell?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the cell emf for the following reaction: Cu2+(0.10 M) + H2(1 atm) Cu(s) + 2H+(pH = 3.00)
A) 0.49 V
B) 0.19 V
C) 0.15 V
D) 0.40 V
E) -0.34 V
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Complete and balance the following redox reaction under basic conditions: PO33-(aq)+ MnO4-(aq) PO43-(aq)+ MnO2(s)
Correct Answer

verified
3PO33-(a...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Many different ways have been proposed to make batteries.One cell is set up with copper and lead electrodes in contact with CuSO4(aq)and Pb(NO3)2 (aq), respectively.If the Pb2+ and Cu2+ concentrations are each 1.0 M, what is the overall cell potential? 
Correct Answer

verified
Correct Answer
verified
Short Answer
Gold can be electrochemically "plated" onto a less expensive metal by submerging the object in a solution of gold(III)and applying an electric current to drive the following half-reaction: Au3+ + 3e- Au(s) How many grams of gold will be formed if a current of 2.37 A is applied for 71 minutes?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reaction Hg2+(aq) + Hg(l) 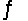 Hg22+(aq) . Find the equilibrium constant for this reaction at 25°C.
Hg22+(aq) . Find the equilibrium constant for this reaction at 25°C.
A) 2 101
B) 6 1059
C) 4 10-3
D) 7 10-2
E) 2 102
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Showing 121 - 138 of 138
Related Exams