B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following liquids is likely to have the highest surface tension?
A) Br 2
B) C 8H 18
C) CH 3OCH 3
D) CH 3OH
E) Pb
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pairs is arranged with the particle of higher polarizability listed first?
A) CCl 4, CI 4
B) H 2O, H 2Se
C) C 6H 14, C 4H 10
D) NH 3, NF 3
E) None of these choices are correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following should have the lowest boiling point?
A) C 5H 12
B) C 6H 14
C) C 8H 18
D) C 10H 22
E) C 12H 26
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The highest temperature at which superconductivity has been achieved is approximately
A) 4 K.
B) 30 K.
C) 70 K.
D) 100 K.
E) 130 K.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
A single water molecule can participate in at most two hydrogen bonds at any instant.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The coordination number of sodium and chloride ions in the NaCl lattice, are, respectively
A) 10 and 10
B) 8 and 8
C) 6 and 6
D) 4 and 4
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In cubic closest packing, the unit cell is body-centered cubic.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 5.00 g sample of water vapor, initially at 155°C is cooled at atmospheric pressure, producing ice at −55°C. Calculate the amount of heat energy lost by the water sample in this process, in kJ. Use the following data: specific heat capacity of ice is 2.09 J/g·K; specific heat capacity of liquid water is 4.18 J/g·K; specific heat capacity of water vapor is 1.84 J/g·K; heat of fusion of ice is 336 J/g; heat of vaporization of water is 2260 J/g.
A) 15.6 kJ
B) 10.2 kJ
C) 5.4 kJ
D) 3.2 kJ
E) 1.6 kJ
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the adhesive forces between a liquid and the walls of a capillary tube are greater than the cohesive forces within the liquid
A) the liquid level in a capillary tube will rise above the surrounding liquid and the surface in the capillary tube will have a convex meniscus.
B) the liquid level in a capillary tube will rise above the surrounding liquid and the surface in the capillary tube will have a concave meniscus.
C) the liquid level in a capillary tube will drop below the surrounding liquid and the surface in the capillary tube will have a convex meniscus.
D) the liquid level in a capillary tube will drop below the surrounding liquid and the surface in the capillary tube will have a concave meniscus.
E) None of these choices are correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Crystal structures may be conveniently measured using
A) X-ray diffraction.
B) infrared spectroscopy.
C) ultraviolet-visible spectroscopy.
D) microwave spectroscopy.
E) magnetic resonance imaging.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following quantities is generally not obtainable from a single heating or cooling curve of a substance, measured at atmospheric pressure?
A) Melting point
B) Boiling point
C) Triple point
D) Heat of fusion
E) Heat of vaporization
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pairs is arranged with the particle of higher polarizability listed first?
A) Se 2−, S 2−
B) I, I −
C) Mg 2+, Mg
D) Br, I
E) None of these choices are correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following phase diagram and identify the process occurring as one goes from point C to point D. 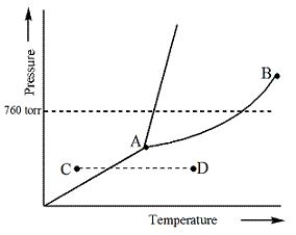
A) Increasing temperature with a phase change from solid to liquid
B) Increasing temperature with a phase change from solid to vapor
C) Increasing temperature with a phase change from liquid to vapor
D) Increasing temperature with no phase change
E) Increasing temperature beyond the critical point
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following properties measures the energy needed to increase the surface area of a liquid?
A) Capillary action
B) Surface tension
C) Viscosity
D) Cohesion
E) Specific elasticity
G) A) and D)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
When silicon is doped with an element from group 3A(13) , the device/material produced is a/an
A) intrinsic semiconductor.
B) p-type semiconductor.
C) n-type semiconductor.
D) p-n junction.
E) transistor.
G) D) and E)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Acetic acid has a heat of fusion of 10.8 kJ/mol and a heat of vaporization of 24.3 kJ/mol. Estimate the value for the heat of sublimation.
A) 35.1 kJ/mol
B) 13.5 kJ/mol
C) −13.5 kJ/mol
D) −35.1 kJ/mol
E) None of these choices are correct
G) B) and E)
Correct Answer

verified
A
Correct Answer
verified
True/False
In the packing of identical atoms with cubic unit cells, the packing efficiency increases as the coordination number increases.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
In a transistor, the current through one semiconductor junction controls the current through a neighboring junction.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about the packing of monatomic solids with different unit cells is incorrect?
A) The coordination number of atoms in hcp and fcc structures is 12.
B) The coordination number of atoms in simple cubic structures is 6.
C) The coordination number of atoms in bcc structures is 8.
D) A bcc structure has a higher packing efficiency than a simple cubic structure.
E) A bcc structure has a higher packing efficiency than a fcc structure.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 95
Related Exams