A) Hg 2+( aq)
B) Cu( s)
C) Cu 2+( aq)
D) Hg( l)
E) Hg 2+( aq) and Cu 2+( aq)
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reaction: 3Co2+(aq) + 6NO3(aq) + 6Na+(aq) + 2PO43-(aq) → Co3(PO4) 2(s) + 6Na+(aq) + 6NO3(aq) Identify the net ionic equation for this reaction.
A) Na +( aq) + NO 3 −( aq) → NaNO 3( aq)
B) 3Co 2+( aq) + NO 3 −( aq) + Na +( aq) + 2PO 4 3−( aq) → Co 3(PO 4) 2( s) + NaNO 3( aq)
C) 3Co 2+( aq) + 6NO 3 −( aq) + 6Na +( aq) + 2PO 4 3−( aq) → Co 3(PO 4) 2( s) + 6NaNO 3( aq)
D) 3Co 2+( aq) + 2PO 4 3−( aq) → Co 3(PO 4) 2( s)
E) None of these choices are correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
1.0M aqueous solutions of the following substances are prepared. Which one would you expect to have the lowest electrical conductivity?
A) NaOH
B) CH 3CH 2OH (ethanol)
C) KBr
D) CH 3COOH (acetic acid)
E) HClO 4
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
All acid-base reactions produce a salt and water as the only products.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sodium thiosulfate, Na2S2O3, is used as a "fixer" in black and white photography. Identify the reducing agent in the reaction of thiosulfate with iodine. 2S2O32-(aq) + I2(aq) → S4O62-(aq) + 2I-(aq)
A) I 2( aq)
B) I −( aq)
C) S 2O 3 2−( aq)
D) S 4O 6 2−( aq)
E) S 2O 3 2−( aq) and I −( aq)
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of H+(aq) ions are present in 1.25 L of 0.75 M nitric acid?
A) 0.60 mol
B) 0.75 mol
C) 0.94 mol
D) 1.7 mol
E) 1.9 mol
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
During a titration, the following data were collected. A 25 mL portion of an unknown acid solution was titrated with 1.0 M NaOH. It required 65 mL of the base to neutralize the sample. How many moles of acid are present in 3.0 liters of this unknown solution?
A) 2.6 moles
B) 0.9 moles
C) 8.7 moles
D) 7.8 moles
E) 5.6 moles
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The oxidation numbers of P, S, and Cl in H2PO2-, H2S, and KClO4 are, respectively
A) −1, −1, +3.
B) −1, −2, +7.
C) +1, −2, +7.
D) −1, −2, +3.
E) +1, +2, +7.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calcium chloride is used to melt ice and snow on roads and sidewalks and to remove water from organic liquids. Calculate the molarity of a solution prepared by diluting 165 mL of 0.688 M calcium chloride to 925.0 mL.
A) 3.86 M
B) 0.743 M
C) 0.222 M
D) 0.123 M
E) 0.114 M
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the classification for the following reaction. 2NaCl(l) 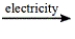 2Na(l) + Cl2(g)
2Na(l) + Cl2(g)
A) Acid-base
B) Precipitation
C) Combination
D) Displacement
E) Decomposition
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aqueous potassium iodate (KIO3) and potassium iodide (KI) react in the presence of dilute hydrochloric acid (HCl) , as shown below. KIO3(aq) + 5KI(aq) + 6HCl(aq) → 3I2(aq) + 6KCl(aq) + 3H2O(l) What mass of iodine (I2) is formed when 15.0 mL of 0.0050 M KIO3 solution reacts with 30.0 mL of 0.010 M KI solution in the presence of excess HCl?
A) 0.023 g I 2
B) 0.029 g I 2
C) 0.057 g I 2
D) 0.046 g I 2
E) 0.076 g I 2
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 111 of 111
Related Exams