A) I < II < III
B) II < I < III
C) I < III < II
D) II < III < I
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following structures contains a primary amine?
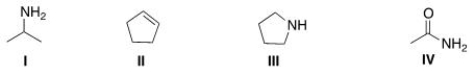
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following structures contains an amide?
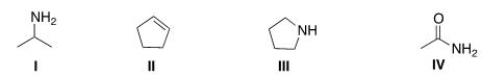
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why do heteroatoms confer reactivity on a particular molecule?
A) Because they have lone pairs and create electron-rich sites on carbon.
B) Because they have lone pairs and create electron-deficient sites on carbon.
C) Because they are electronegative and act as electrophiles.
D) Because they are electropositive and act as nucleophiles.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What molecular features are required for soap to properly dissolve grease and oil?
A) The molecule must be large.
B) The molecule must contain a polar head.
C) The molecule must contain a non-polar tail.
D) The molecule must contain both a polar head and a non-polar tail.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following intermolecular forces would not form between similar molecules of the structure below?
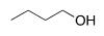
A) London dispersion forces
B) Ion-ion
C) Hydrogen bonding
D) Dipole-dipole
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following alkanes is expected to have the highest melting point?
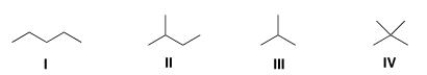
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is expected to be H2O soluble?
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds has the highest boiling point?

A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What intermolecular force is generally considered the strongest?
A) Hydrogen bonding
B) London dispersion forces
C) Covalent bonds
D) Dipole-dipole
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
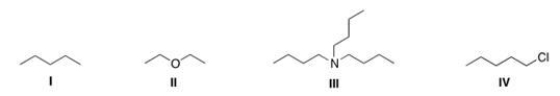
Rank the following compounds in order of increasing strength of intermolecular forces. 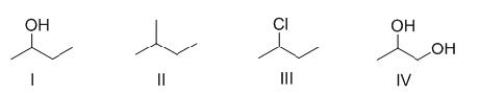
A) I > III > IV > II
B) IV > II > I > III
C) IV > I > III > II
D) I > IV > II > III
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is expected to be the least soluble in H2O?

A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The indicated bond is:
A) Nucleophilic because it is electron-deficient.
B) Electrophilic because it is electron-deficient.
C) Nucleophilic because it electron-rich.
D) Electrophilic because it is electron-rich.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of decreasing melting point, putting the compound with the highest melting point first. 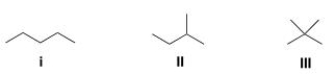
A) I > II > III
B) II > III > I
C) III > II > I
D) III > I > II
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
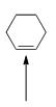
Which of the following molecules are aromatic hydrocarbons?
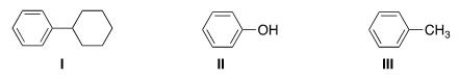
A) I
B) II
C) III
D) I and III
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about the solubility of organic compounds in H2O is true?
A) The non-polar part of a molecule that is not attracted to water is said to be hydrophilic.
B) The non-polar part of a molecule that is not attracted to water is said to be hydrophobic.
C) The polar part of a molecule that can that can bond hydrogen to water is said to be hydrophobic.
D) For an organic compound with one functional group that contains an O or N atom, the compound is water soluble only if it has 35 carbons.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following lists the correct functional groups found in aspartame, the artificial sweetener?
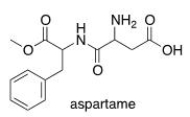
A) Amine, aromatic, carboxylic acid, ether, ketone.
B) Amine, amide, aromatic, carboxylic acid, ester.
C) Amide, alcohol, aromatic, carboxylic acid, ether.
D) Amine, aromatic, carboxylic acid, ester, nitrile.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds has the highest boiling point?
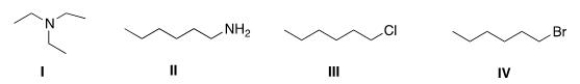
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules contain the same functional groups?

A) I, II, IV
B) I, II, III
C) II, III, IV
D) I, III, IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a secondary alcohol?

A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 45
Related Exams