A) calcium dichloride
B) dicalcium trichloride
C) dicalcium chloride
D) calcium chloride
E) dicalcium dichloride
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the total number of valence electrons in the chloroethylene molecule (C2H3Cl) ?
A) 6
B) 18
C) 21
D) 32
E) 62
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A compound contains magnesium and phosphate. What is the formula unit of this compound?
A) Mg3P2
B) MgPO4
C) Mg3(PO4) 2
D) Mg2(PO4) 3
E) MgHPO4
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Chloroform (CHCl3) is an anesthetic and is also used in the synthesis of ozone damaging refrigerants called CFCs. Which of the following is the Lewis structure for chloroform?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a diatomic molecule?
A) N2
B) NaCl
C) H2O
D) CO
E) Li2O
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In addition to single bonds, which of the following electron groups are on the nitrogen of methylamine (CH5N) ?
A) a double bond
B) a triple bond
C) a single electron
D) a lone pair of electrons
E) None of the above
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the molecules below contain an atom with an expanded octet? 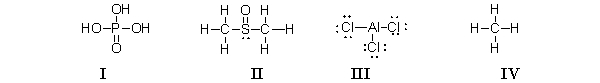
A) I and II
B) III only
C) IV only
D) I, II, and III
E) All of these molecules have an expanded octet.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the charge on iron in the ionic compound FeCl3?
A) +3
B) +1
C) 0
D) (1)
E) (3)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements is the least electronegative?
A) potassium
B) hydrogen
C) fluorine
D) carbon
E) oxygen
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Tamoxifen has some key similarities to estradiol. Which of the following statements describes one of the similarities?
A) Tamoxifen has the same structure as estradiol.
B) Tamoxifen has the same number of atoms as estradiol.
C) Tamoxifen performs the same function in the cell as estradiol.
D) Tamoxifen binds to the same receptor as estradiol.
E) Tamoxifen deforms the shape of the estrogen receptor in the same way as estradiol.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the electron geometry of the carbon and the nitrogen in methylamine (CH5N) ?
A) Nitrogen is trigonal planar and carbon is tetrahedral.
B) Carbon is tetrahedral and nitrogen is bent.
C) Nitrogen is trigonal pyramidal and carbon is tetrahedral.
D) Carbon is linear and nitrogen is tetrahedral.
E) Both of them are tetrahedral.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the bonds in the following molecule are polar covalent? 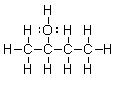
A) C-H and O-H only
B) C-O and O-H only
C) C-O only
D) C-C and C-O
E) O-H only
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many atoms are bonded to the carbon in dichloromethane (CH2Cl2) ?
A) 0
B) 1
C) 2
D) 3
E) 4
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Is chloroform (CHCl3) a polar molecule?
A) No, all polarities cancel out.
B) Yes, all molecules with polar bonds are polar.
C) No, the bonds in chloroform are not polar.
D) Yes, the polarities do not cancel out.
E) No, carbon compounds cannot be polar.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the ratio of ions in this lattice of lithium (the larger spheres) and oxygen (the smaller spheres) in Li2O? 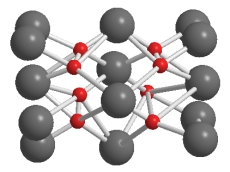
A) 1 Li:1 O
B) 2 Li:1 O
C) 2 O:1 Li
D) 4 O:5 Li
E) 4 Li:5 O
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What does a ball-and-stick model show clearly that a space-filling model does not?
A) relative atom size
B) relative bond length
C) relative bond strength
D) bond angle
E) atom identity
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Electrons shared between two atoms is a(n) _______.
A) cation
B) anion
C) ionic bond
D) covalent bond
E) salt
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Electronegativity is a measure of an atom's ability to
A) become an anion.
B) ionize.
C) draw electrons to itself in a covalent bond.
D) accept an electron from a metal.
E) donate an electron to a nonmetal.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A polar molecule is one that has
A) one large atom and one small atom.
B) an even distribution of charge.
C) a positive or negative charge.
D) a more positive side and a more negative side.
E) only polar atoms.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following covalent bonds is polar?
A) C-C
B) C-H
C) O-Cl
D) Cl-Cl
E) Mg-Cl
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 164
Related Exams