A) 2.16 × 1014 kJ
B) 2.16 × 1011 kJ
C) 2.16 × 108 kJ
D) 2.16 × 105 kJ
E) None of these choices are correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the missing species in the following nuclear transmutation. 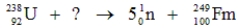
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the nuclide that completes the following nuclear reaction. 
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Who discovered radioactivity?
A) Geiger
B) Curie
C) Roentgen
D) Becquerel
E) Rutherford
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A pure sample of tritium, 3H, was prepared and sealed in a container for a number of years. Tritium undergoes β decay with a half-life of 12.32 years. How long has the container been sealed if analysis of the contents shows there are 5.25 mol of 3H and 6.35 mol of 3He present?
A) 2.34 y
B) 3.38 y
C) 9.77 y
D) 14.1 y
E) 25.6 y
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isotope 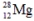 has a half-life of 21 hours. If a sample initially contains exactly 10000 atoms of
has a half-life of 21 hours. If a sample initially contains exactly 10000 atoms of 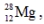 approximately how many of these atoms will remain after one week?
approximately how many of these atoms will remain after one week?
A) 1250
B) 78
C) 39
D) 0
E) None of these choices are correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following descriptions relating to nuclear reactions is correct?
A) The ratio of neutrons to protons remains constant.
B) The number of protons plus neutrons remains constant.
C) The number of electron remains constant.
D) The total charge changes.
E) The total number of nucleons changes.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Gamma rays are not deflected by an electric field.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The radioisotope 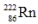 will decay through
will decay through
A) α decay.
B) β decay.
C) ![]() decay.
decay.
D) positron decay.
E) electron capture.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Assuming that no other particles are produced, which of the following particles could be used to bombard nitrogen-14 in order to make fluorine-18?
A) alpha particle
B) beta particle
C) neutron
D) proton
E) positron
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The difference between the rad and the rem is
A) the rem is a rad per year.
B) the rad takes into account the type of radiation.
C) the rem takes into account the effect on the particular biological tissue.
D) the rem is a rad per kilogram.
E) None of these choices are correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Detection of radiation by a Geiger-Müller counter depends on
A) the emission of a photon from an excited atom.
B) the ability of an ionized gas to carry an electrical current.
C) the emission of a photon of light by the radioactive particle.
D) the ability of a photomultiplier tube to amplify the electrical signal from a phosphor.
E) the detection of the sound made by decay particles.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A N-14 nucleus is hit by a particle, forming a C-14 nucleus and a proton as the only products. Identify the type of particle which struck the N-14 nucleus.
A) alpha
B) proton
C) electron
D) neutron
E) deuterium
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Exposure to 10 nCi for 10 minutes is more hazardous for a child than for an adult because
A) the child's cells are dividing more rapidly than the adult's and are, therefore, more susceptible to the radiation.
B) the child's smaller body size makes the effective dose larger for the child than for the adult.
C) the child's immune system is not developed well enough to resist damage.
D) the child's skin is not as thick as an adult's and cannot block as much radiation.
E) None of these choices are correct.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The (negative) binding energy per nucleon reaches a maximum for the isotope 126C
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the nuclide that completes the following nuclear reaction. 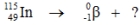
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The nuclide Pb-210 undergoes three successive decays (beta, alpha, and beta, respectively) to form a stable nuclide. What are the three nuclides that form from Pb-210 in this decay series?
A) Tl-210, Au-206, Pt-206
B) Bi-210, Tl-206, Pb-206
C) Pb-209, Hg-205, Hg-204
D) Bi-210, Pb-206, Bi-206
E) None of these choices are correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is an incorrect representation of the indicated particle or nucleus?
A) positron: ![]()
B) neutron: ![]()
C) helium-3: ![]()
D) alpha particle: ![]()
E) proton: ![]()
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following series of radioactive decays would convert Pa-234 to Ra-226?
A) beta, alpha, beta
B) alpha, alpha
C) beta, alpha, alpha, beta
D) beta, alpha, alpha
E) alpha, beta, gamma
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 30.0-kg child receives 2.65 × 107 β particles, each with an energy of 4.60 × 10-13 J. If the RBE = 0.78, how many millirem did the child receive?
A) 3.2 × 10-7
B) 5.2 × 10-7
C) 5.2 × 10-4
D) 3.2 × 10-2
E) None of these choices are correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 81
Related Exams