A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 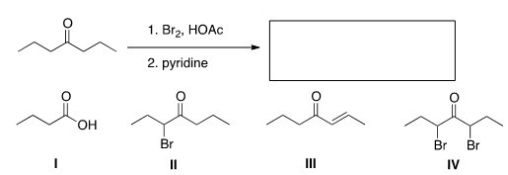
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds would undergo racemization in the presence of a base? 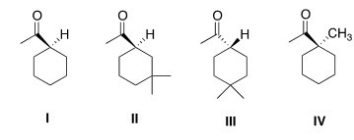
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the least acidic compound? 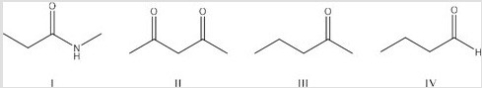
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Will acetone be completely deprotonated by potassium tert-butoxide?
A) Yes
B) No
D) undefined
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is the enolate of acetone less basic than the allyl anion derived from propene?
A) Because there are more atoms in acetone
B) Because there are more resonance structures for the enolate of acetone
C) It isn't; the allyl anion is less basic.
D) One of the resonance structures for the enolate places the negative charge on the more electronegative oxygen.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A simple chemical test to distinguish between acetone and 3-pentanone would be the reaction of the compounds with
A) bromine with acetic acid.
B) bromine and aqueous hydroxide ion.
C) THF,LDA at -78 °C followed by reaction with bromine.
D) base and methyl bromide.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 47 of 47
Related Exams